UK MS Register (Swansea University)
Charity / Academia
 The UK MS Register is an innovative research study designed to increase our understanding of living with Multiple Sclerosis (MS) in the UK.
The UK MS Register is an innovative research study designed to increase our understanding of living with Multiple Sclerosis (MS) in the UK.
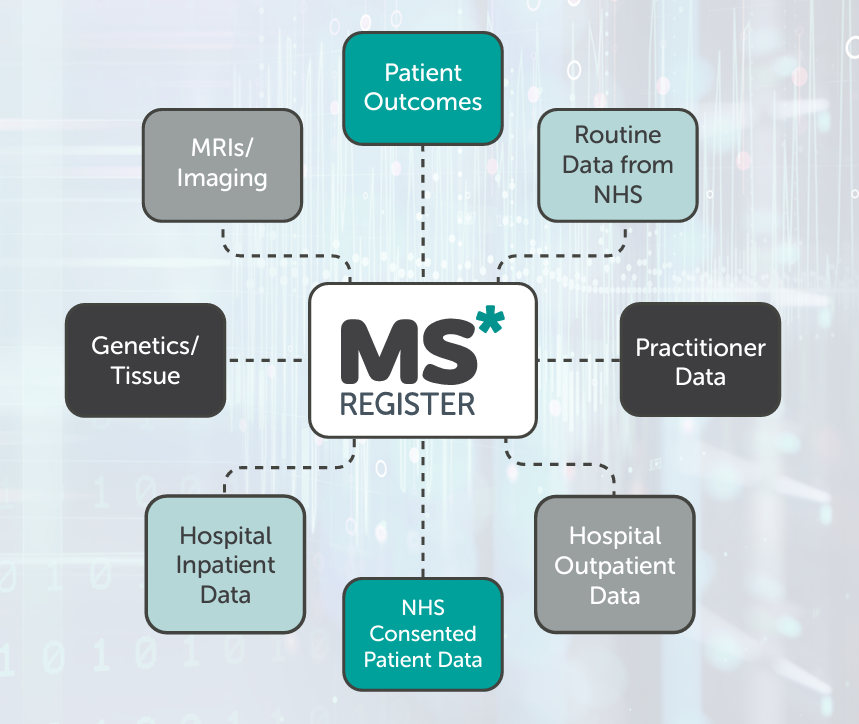
It is estimated that more than 130,000 people in the UK live with MS. The Register supports research into this condition by collecting data directly from people with MS and from the NHS.
Data is gathered from:
- People with MS sharing information about their experiences directly with the Register in the form of Patient/Participant Reported Outcomes (PROs).
- Collaboration with NHS Trusts across the UK to securely transmit appropriately consented people with MS’ medical records.
Since 2011 the Register has collected data about the physical, environmental and social impact on people living with MS. The Register now holds one of the largest repositories of ‘real world’ data about the disease, opening up huge opportunities for research.
The data is used by the Register team at Swansea University, but is also available through simple governance application across the UK and the World.
There have been individually funded studies by Merck KgAA, Sanofi, Biogen Idec and Novartis.
View the UK MS Register’s recent publications
The UK MS Register is based in Swansea University Medical School and is funded by the MS Society, who have awarded the Register a total of approximately £10 million over 10 years.
How to Work with the UK MS Register
The Register holds a vast array of real world data collected directly from people with MS and the NHS. There is a clinical minimum data set [Middleton et al 2017] and people with MS directly supply data using PROs such as the Expanded Disability Status Scale, Hospital Anxiety and Depression Scale, Multiple Sclerosis Walking Scale, Fatigue Severity Scale, EuroQoL EQ-5D 3L/5l and the Multiple Sclerosis Impact Scale 29. Additionally there are systematic regular collections of epidemiological, demographic and treatment data. We are regularly contacted by researchers from across the globe who are keen to access this impressive resource.
You can apply to conduct your research either using data we already hold, by hosting a questionnaire on our web platform, or by conducting recruitment to a clinical trial.
